
Ostomy Supporting Products – Information
Disclaimer: The information in this post is based on product knowledge from product information direct from the manufacturer and also personal use of products within my own stoma care. All experiences given here are my own and should not be taken as medical advice. If you require advice or support about any aspect of your stoma care, please contact your SCN in the first instance or your GP.
If you are looking for the entire ‘Ostomy Supporting Products‘ series, you can find them all here.
In previous posts in this series, I’ve explained how to use individual products as well as when to use them. This is called ‘best practice‘ and is something that a Stoma Care Nurse (SCN) should advise you on, if you require any of the individual supporting products.
Whilst ostomy supplies in the UK are covered by a ‘exemption’ certificate – either as a Prepayment Certificate (see conditions here) or as a Medical Exemption Certificate (see conditions here) – there is still a cost. This cost is covered by GP practices, which are governed and overseen by Clinic Comissioning Groups (CCG’s). These CCG’s hold the purse strings of an entire geographical location for all GP surgeries within that area.
And they all rule differently. This is not exclusive to just ostomy products, but for all items which require a prescription to dispense. For some CCG’s, they simplify what can and can not be dispensed by using a Formulary. This document dictates the specific products they are most likely to agree for use and the qualities in which they will dispense. To make it fair, they treat every patient who has a stoma exactly the same.
I currently reside within the Cambridgeshire and Peterborough CCG area and their view is:
“The Cambridgeshire and Peterborough Formulary has been designed to assist in promoting safe, effective and economic prescribing across the Cambridgeshire and Peterborough health community. It comprises a list of medicines with guidance on locally recommended drug choices. The content has been agreed by the Cambridgeshire and Peterborough Joint Prescribing Group and by the provider trusts’ Drugs and Therapeutics Committees.
Prescribers in primary care should select ‘green traffic light status’ medicines from the formulary whenever possible, for treatment initiation and where treatments are being reviewed.”
That is not to say that you can not have these restrictions overruled. This is a process which can take place, with usually the advice and recommendation of your SCN speaking to your GP surgery. However, the GP surgery still will have the ability to overrule a decision if the patient overstocks products are is misusing them. There are guidelines for these instances on all individual CCG’s NHS websites.
You can find out your own local CCG but searching here.
Guidance on Products and Usage on Prescribing
Below are tables of an average overview of what a CCG will see as acceptable usage and dispensing of products. The one below begins with stoma bags and moved on to supporting products.
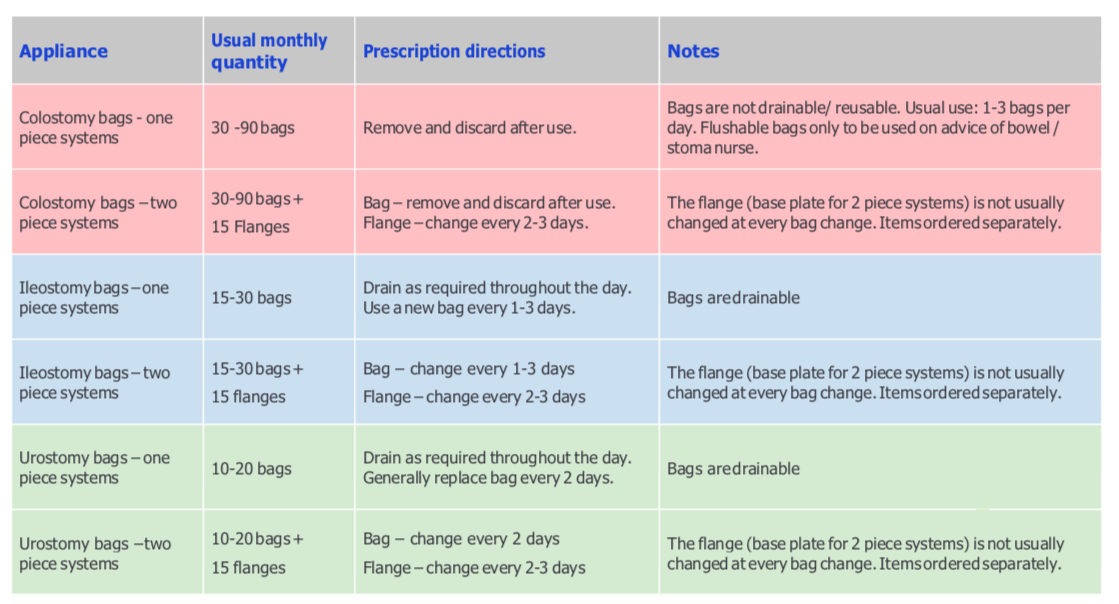


For some, these recommendations and guidelines do not correlate with their personal circumstances. However, it is stated that “Prescribing stoma appliances – quantities and appliance choice will be made based on the individual needs of the patient” which comes from the guidance of your SCN’s assessment of your needs when seeing you in clinic or on a home visit. There are good ways to ensure continuation of care and the proper usage of supplies.
But as some times thing do change with your stoma, it is the patient’s responsibility – with recommendations from your SCN – to make your changes known, stated here: “if changes are made to the patient’s prescription then the prescriber and GP practice and dispensing appliance or pharmacy contractor must be informed at the earliest opportunity and all details must be documented in the notes.”
Prescribers do have a right to enquire about your usage. And whilst it’s sometimes very difficult or near impossible to second guess your usage at times, it is best to be up front and honest. At most GP’s you can request a call back and speak to someone to have your products and usages documents on your medical notes. As well as having a yearly review of medications, there is also a drive for this to be applied to appliances too.
Recommended Interventions:
Circumstances that may require a GP / prescriber referral to stoma care specialist:
- Routine over ordering of stoma supplies.
- Long term use ≥ 3 months of skin protective products (wipes / films / paste / powders).
- Current use of pressure plates or shields – patient may benefit from the use of newer products with built in convexity.
- Old style reusable bags.
- Current use of adhesive rings, discs pads or plasters – newer products may be more appropriate.
- Current use of products that are to be discontinued.
- Patients that are experiencing leakage.
- Patients experiencing dietary problems.
- Patients that have developed hernias.
Things to note:
- Upon having a stoma, you should be told how long this will be – ie either temporary for less that 6 months or permanent – so your SCN can determine which exemption you fall into. My exemption was done for me whilst in recovery from my surgery and my card turned up a couple of days after being discharged home. If you do not get this done for you, you can apply here but please read the conditions that come with it. Not everything will be covered, so it’s helpful to know where you stand.
- There is nothing to say you can’t have some thing, if you can prove it’s use. For some, they find this another hardship to bear but this is why SCN’s can help! Even making an appointment to discuss things with your GP can go along way. Being open to communication and will to express why you need something, instead of wanting it, can help too.
- If you’re struggling with any of your supplies or how to use them, it is best to speak directly to a SCN in the first instance or a care line which can explain how and when to use products. Some DAC’s offer this service under their guidance within the NHS (see ‘Overview of Service Provision’ 4.2)
- In some CCG’s they require the patient or direct relation response for their prescriptions to order items themselves, or on their behalf. This means that only the patient can request products when required; not by anyone else – for example a DAC as a third-party or a pharmacy as part of repeat dispensing. This keeps the CCG’s informed of usage and responsible prescribing.
- The aim for the above note is “to ensure that patients obtain repeat supplies of clinically necessary incontinence &/or stoma products in a timely manner. Whilst also reducing inappropriate or duplicate requests for supply & therefore reducing avoidable waste.”
NB: please note, things will vary between CCG’s. If you are concerned about your products, your usage or anything being ‘rejected / cancelled’ please speak directly to your GP practice’s prescribing team.
Do you have any questions or queries? Or just want to share your own experiences? You can leave me a reply here or leave comments via my social media accounts – on Twitter, find my blog page on Facebook and over on Instagram

Sources:
Cambridgeshire and Peterborough Clinical Commissioning Group formulary and Drug classification: (under subsection of ‘Continence‘):
Further Reading:
Hollister US – Ostomy Accessories: Necessary Tools or Nice to Have Options?
Association of Stoma Care Nurses UK – ASCN Stoma Care: National Clinical Guidelines
SecuriCare Medical – Stoma Care Clinical Nursing Standards (RCN Accredited)



6 Comments
Assumptions About IBD & Stomas •
26/02/2021 at 19:02
[…] If you enjoyed this post check out Being and Staying Positive and What Advice do Patients look for Prior to Ostomy Surgery […]
"How different are scans when you have a bag?" •
11/03/2021 at 20:48
[…] If you enjoyed this post check out Ostomy Glossary and Ostomy Supporting Products – Information […]
"Will you always need check-ups for your IBD?" •
27/03/2021 at 14:58
[…] If you enjoyed this post check out Ostomy Glossary and Ostomy Supporting Products – Information […]
What is Convexity? •
03/04/2021 at 21:15
[…] you enjoyed this post check out The Importance of a AUR – Applicance User Review , Ostomy Supporting Products – Information and Open Letter: Stoma Bag […]
"Does caffeine affect your IBD?" •
23/06/2021 at 16:47
[…] If you enjoyed this post check out Fibre with IBD and The What and Why: Dehydration […]
"What do gelling agents do?" •
23/06/2021 at 16:48
[…] If you enjoyed this post check out Ostomy Glossary and Ostomy Supporting Products – Information […]